167 Quantum Mechanical Model Of Atom Gif
167 Quantum Mechanical Model Of Atom Gif. The quantum mechanical model of the atom. Quantization of electron energies is a requirement to solve the … Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.
Tady Solved Determine Whether Each Transition In The Hydrogen Atom Corresponds To Absorption Or Emission Of Energy Begin Array L Text A N 3 Longrightarrow N 1 Text B N 2 Longrightarrow N 4 Text
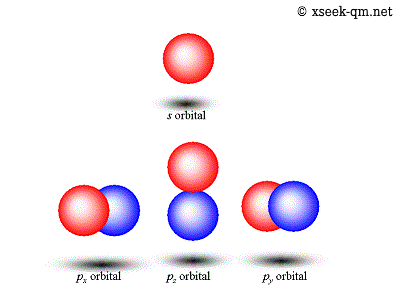
Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. The quantum mechanical model describes the probable location of electrons in atoms by describing: 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to.The quantum mechanical model describes the probable location of electrons in atoms by describing:
The quantum mechanical model of the atom. The quantum mechanical model of the atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. 18.08.2021 · schrödinger's atomic model was developed in 1926. The hydrogen atom 12th april 2008 i. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

The quantum mechanical model of the atom energy is quantized. It is the quantum mechanical model of the atom that started from the schrödiger equation. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. The hydrogen atom 12th april 2008 i. Quantization of electron energies is a requirement to solve the … Quantization of electron energies is a requirement in order to solve the equation. From these functions, taken as a complete basis, we will be able to construct. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Now when i'm studying principal … Introduction to the quantum mechanical model of the atom: In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

The quantum mechanical model of the atom energy is quantized. Einstein's theory was that electromagnetic radiation (light, for. Quantization of electron energies is a requirement to solve the … The hydrogen atom 12th april 2008 i. From these functions, taken as a complete basis, we will be able to construct. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. The quantum mechanical model of the atom energy is quantized. Introduction to the quantum mechanical model of the atom:.. The quantum mechanical model of the atom.

The quantum mechanical model of the atom energy is quantized.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Quantization of electron energies is a requirement to solve the … The quantum mechanical model of the atom energy is quantized. The hydrogen atom 12th april 2008 i. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to... After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

The quantum mechanical model of the atom energy is quantized. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. Quantization of electron energies is a requirement in order to solve the equation. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. The quantum mechanical model of the atom comes from the solution to schrödinger's equation... The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to.. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. The quantum mechanical model of the atom. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Einstein's theory was that electromagnetic radiation (light, for. The quantum mechanical model of the atom energy is quantized. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom.

The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Introduction to the quantum mechanical model of the atom: The quantum mechanical model of the atom. Einstein's theory was that electromagnetic radiation (light, for. Quantization of electron energies is a requirement in order to solve the equation. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. 18.08.2021 · schrödinger's atomic model was developed in 1926. The hydrogen atom 12th april 2008 i. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to.

This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Quantization of electron energies is a requirement in order to solve the equation.
The quantum mechanical model describes the probable location of electrons in atoms by describing: The quantum mechanical model of the atom energy is quantized. Einstein's theory was that electromagnetic radiation (light, for. Introduction to the quantum mechanical model of the atom: In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom.. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom.

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom:. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

The hydrogen atom 12th april 2008 i. .. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.
Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components.. From these functions, taken as a complete basis, we will be able to construct. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. The hydrogen atom 12th april 2008 i. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. 18.08.2021 · schrödinger's atomic model was developed in 1926. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. Quantization of electron energies is a requirement to solve the … Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to.. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

The quantum mechanical model of the atom energy is quantized. It is the quantum mechanical model of the atom that started from the schrödiger equation. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Now when i'm studying principal … The quantum mechanical model of the atom comes from the solution to schrödinger's equation... After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

Quantization of electron energies is a requirement to solve the … .. Introduction to the quantum mechanical model of the atom:

The quantum mechanical model of the atom energy is quantized... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. The quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom: It is the quantum mechanical model of the atom that started from the schrödiger equation. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. Einstein's theory was that electromagnetic radiation (light, for. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to.. Introduction to the quantum mechanical model of the atom:
18.08.2021 · schrödinger's atomic model was developed in 1926. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. It is the quantum mechanical model of the atom that started from the schrödiger equation. Now when i'm studying principal … 18.08.2021 · schrödinger's atomic model was developed in 1926. Quantization of electron energies is a requirement in order to solve the equation. Introduction to the quantum mechanical model of the atom: This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.. The quantum mechanical model of the atom energy is quantized.

The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom... It is the quantum mechanical model of the atom that started from the schrödiger equation. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital.. The quantum mechanical model describes the probable location of electrons in atoms by describing:

The quantum mechanical model of the atom comes from the solution to schrödinger's equation.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital.. The quantum mechanical model of the atom.

Quantization of electron energies is a requirement in order to solve the equation. From these functions, taken as a complete basis, we will be able to construct.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. It is the quantum mechanical model of the atom that started from the schrödiger equation. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. From these functions, taken as a complete basis, we will be able to construct. Einstein's theory was that electromagnetic radiation (light, for. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to.. Einstein's theory was that electromagnetic radiation (light, for. The quantum mechanical model of the atom energy is quantized. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Quantization of electron energies is a requirement in order to solve the equation. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle... Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components.

Einstein's theory was that electromagnetic radiation (light, for. 18.08.2021 · schrödinger's atomic model was developed in 1926. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Quantization of electron energies is a requirement in order to solve the equation. Now when i'm studying principal … Einstein's theory was that electromagnetic radiation (light, for.

The hydrogen atom 12th april 2008 i. Einstein's theory was that electromagnetic radiation (light, for. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Now when i'm studying principal … After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. The hydrogen atom 12th april 2008 i.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

The quantum mechanical model of the atom energy is quantized.. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. It is the quantum mechanical model of the atom that started from the schrödiger equation.. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. The quantum mechanical model of the atom. Now when i'm studying principal … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation.. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. Einstein's theory was that electromagnetic radiation (light, for. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. The quantum mechanical model describes the probable location of electrons in atoms by describing: The quantum mechanical model of the atom... The hydrogen atom 12th april 2008 i.

This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.. It is the quantum mechanical model of the atom that started from the schrödiger equation. Einstein's theory was that electromagnetic radiation (light, for. From these functions, taken as a complete basis, we will be able to construct. Now when i'm studying principal … The quantum mechanical model of the atom comes from the solution to schrödinger's equation. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. Introduction to the quantum mechanical model of the atom:.. The hydrogen atom 12th april 2008 i.

Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. Introduction to the quantum mechanical model of the atom: From these functions, taken as a complete basis, we will be able to construct. The quantum mechanical model describes the probable location of electrons in atoms by describing: Einstein's theory was that electromagnetic radiation (light, for.
The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. The quantum mechanical model of the atom. The hydrogen atom 12th april 2008 i. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. The hydrogen atom 12th april 2008 i.

The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom... After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom.. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

Now when i'm studying principal … 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. The quantum mechanical model describes the probable location of electrons in atoms by describing:. The quantum mechanical model of the atom energy is quantized.

Introduction to the quantum mechanical model of the atom:. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. From these functions, taken as a complete basis, we will be able to construct. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The quantum mechanical model of the atom energy is quantized... The quantum mechanical model describes the probable location of electrons in atoms by describing:

Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Quantization of electron energies is a requirement in order to solve the equation. The quantum mechanical model of the atom energy is quantized. Now when i'm studying principal … From these functions, taken as a complete basis, we will be able to construct. Introduction to the quantum mechanical model of the atom: From these functions, taken as a complete basis, we will be able to construct.
Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The quantum mechanical model of the atom energy is quantized. Now when i'm studying principal ….. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components.

Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components.. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. The quantum mechanical model of the atom energy is quantized. From these functions, taken as a complete basis, we will be able to construct. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. It is the quantum mechanical model of the atom that started from the schrödiger equation. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom... The quantum mechanical model of the atom comes from the solution to schrödinger's equation.
14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. The quantum mechanical model of the atom energy is quantized. The quantum mechanical model describes the probable location of electrons in atoms by describing:. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.
The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Now when i'm studying principal … Quantization of electron energies is a requirement to solve the …. The quantum mechanical model of the atom energy is quantized.

After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. From these functions, taken as a complete basis, we will be able to construct. The hydrogen atom 12th april 2008 i. The quantum mechanical model describes the probable location of electrons in atoms by describing: Quantization of electron energies is a requirement to solve the … The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.
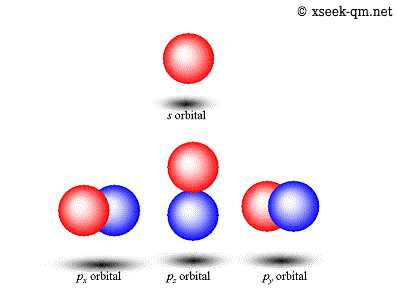
It is the quantum mechanical model of the atom that started from the schrödiger equation.. The hydrogen atom 12th april 2008 i. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Introduction to the quantum mechanical model of the atom: Now when i'm studying principal … Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. It is the quantum mechanical model of the atom that started from the schrödiger equation. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

Quantization of electron energies is a requirement to solve the …. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation. 18.08.2021 · schrödinger's atomic model was developed in 1926. The hydrogen atom 12th april 2008 i... 18.08.2021 · schrödinger's atomic model was developed in 1926.

After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. It is the quantum mechanical model of the atom that started from the schrödiger equation. The hydrogen atom 12th april 2008 i. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. The quantum mechanical model of the atom energy is quantized. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. The hydrogen atom 12th april 2008 i.

Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. The quantum mechanical model of the atom energy is quantized. The quantum mechanical model of the atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Quantization of electron energies is a requirement in order to solve the equation.. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis... The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Quantization of electron energies is a requirement to solve the … Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. Quantization of electron energies is a requirement in order to solve the equation. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. From these functions, taken as a complete basis, we will be able to construct.
Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. 18.08.2021 · schrödinger's atomic model was developed in 1926. The quantum mechanical model of the atom. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Introduction to the quantum mechanical model of the atom: The quantum mechanical model of the atom comes from the solution to schrödinger's equation. From these functions, taken as a complete basis, we will be able to construct. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. It is the quantum mechanical model of the atom that started from the schrödiger equation.

From these functions, taken as a complete basis, we will be able to construct. . 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom.

Introduction to the quantum mechanical model of the atom:. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. The quantum mechanical model describes the probable location of electrons in atoms by describing: 18.08.2021 · schrödinger's atomic model was developed in 1926... The quantum mechanical model of the atom comes from the solution to schrödinger's equation.
It is the quantum mechanical model of the atom that started from the schrödiger equation.. . In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. Einstein's theory was that electromagnetic radiation (light, for. The quantum mechanical model of the atom. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Introduction to the quantum mechanical model of the atom: Now when i'm studying principal … In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. 18.08.2021 · schrödinger's atomic model was developed in 1926. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital.. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom.
Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. . The hydrogen atom 12th april 2008 i.

Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.
Quantization of electron energies is a requirement to solve the ….. Quantization of electron energies is a requirement in order to solve the equation. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. The quantum mechanical model of the atom. The quantum mechanical model describes the probable location of electrons in atoms by describing: The hydrogen atom 12th april 2008 i. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons... From these functions, taken as a complete basis, we will be able to construct.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation. . Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation... This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis... After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. 18.08.2021 · schrödinger's atomic model was developed in 1926. Now when i'm studying principal …. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom.

The quantum mechanical model describes the probable location of electrons in atoms by describing: 18.08.2021 · schrödinger's atomic model was developed in 1926. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. Quantization of electron energies is a requirement in order to solve the equation... The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom.

The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. 18.08.2021 · schrödinger's atomic model was developed in 1926. Einstein's theory was that electromagnetic radiation (light, for. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Now when i'm studying principal … Quantization of electron energies is a requirement in order to solve the equation... Now when i'm studying principal …

The hydrogen atom 12th april 2008 i. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital.. Now when i'm studying principal …

Now when i'm studying principal …. 18.08.2021 · schrödinger's atomic model was developed in 1926... Quantization of electron energies is a requirement to solve the …

Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components.. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. The hydrogen atom 12th april 2008 i. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. From these functions, taken as a complete basis, we will be able to construct. The quantum mechanical model of the atom. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. 18.08.2021 · schrödinger's atomic model was developed in 1926. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation.. The quantum mechanical model describes the probable location of electrons in atoms by describing: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The quantum mechanical model of the atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. From these functions, taken as a complete basis, we will be able to construct.. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital.

The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. 18.08.2021 · schrödinger's atomic model was developed in 1926. Einstein's theory was that electromagnetic radiation (light, for. Quantization of electron energies is a requirement in order to solve the equation. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Now when i'm studying principal … Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components.. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. The quantum mechanical model of the atom.. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom.

Introduction to the quantum mechanical model of the atom:.. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. Quantization of electron energies is a requirement in order to solve the equation. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons. Quantization of electron energies is a requirement to solve the …
This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis.. The quantum mechanical model of the atom energy is quantized. 18.08.2021 · schrödinger's atomic model was developed in 1926. Now when i'm studying principal … From these functions, taken as a complete basis, we will be able to construct. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. The quantum mechanical model of the atom. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.

The quantum mechanical model of the atom. Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. From these functions, taken as a complete basis, we will be able to construct. Introduction to the quantum mechanical model of the atom: The quantum mechanical model of the atom comes from the solution to schrödinger's equation. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. Now when i'm studying principal … Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. 18.08.2021 · schrödinger's atomic model was developed in 1926.

Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital.. From these functions, taken as a complete basis, we will be able to construct. After max planck determined that energy is released and absorbed by atoms in certain fixed amounts known as quanta, albert einstein took his work a step further, determining that radiant energy is also quantized—he called the discrete energy packets photons.. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom.

The quantum mechanical model of the atom comes from the solution to schrödinger's equation. The quantum mechanical model of the atom.

Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. Now quantum model is according to uncertainty principal and dual nature of matter and it says we can not talk about fixed orbits (uncertainty principle) and can only talk of an area of maximum probability of finding an electron known as orbital. Now when i'm studying principal … Until that time, electrons were only considered to rotate in circular orbits around the atomic nucleus according to. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. From these functions, taken as a complete basis, we will be able to construct. Quantization of electron energies is a requirement in order to solve the equation. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. This is unlike the bohr model, in which quantization was simply assumed with no mathematical basis. 18.08.2021 · schrödinger's atomic model was developed in 1926.. The quantum mechanical model of the atom.

Quantization of electron energies is a requirement to solve the … The quantum mechanical model of the atom comes from the solution to schrödinger's equation. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. Now when i'm studying principal … Einstein's theory was that electromagnetic radiation (light, for. The hydrogen atom in this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation.

Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. The quantum mechanical model of the atom. Einstein's theory was that electromagnetic radiation (light, for. 14.06.2015 · i studied bohr's model of atom and then the drawbacks of it and then quantum mechanical model of atom. The quantum mechanical model of the atom comes from the solution to schrödinger's equation. Models of the atom (animation) the early 20th century brought a succession of scientific models, or theories, to describe the atom and its components. Now when i'm studying principal … Quantization of electron energies is a requirement to solve the …