197 Atom Carbon Diagram
197 Atom Carbon Diagram. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). The valency of carbon are 2,4 and valence electrons of carbon is 4. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. This image of a carbon atom is taken from a postsecondary chemistry textbook.
Prezentováno Carbon Dioxide Co2 Atomic Diagram An Atomic Diagram Of Carbon Dioxide Or Co2 Showing Its Protons Neutrons And Electrons Canstock
Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. Electron configuration of carbon is 1s 2 2s 2 2p 2. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented.20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented.
/captionthe image on the left is a basic atom diagram. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. Electron configuration of carbon and full orbital diagram. The valency of carbon are 2,4 and valence electrons of carbon is 4. This one shows the protons, neutrons, and electrons of a carbon atom. This image of a carbon atom is taken from a postsecondary chemistry textbook.

The full page where it appears can be … Electron configuration of carbon and full orbital diagram. The valency of carbon are 2,4 and valence electrons of carbon is 4. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. The full page where it appears can be … If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The symbol of carbon is written as 6. Electron configuration of carbon is 1s 2 2s 2 2p 2. This image of a carbon atom is taken from a postsecondary chemistry textbook.

Each is in a …. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). The valency of carbon are 2,4 and valence electrons of carbon is 4. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Each is in a ….. This one shows the protons, neutrons, and electrons of a carbon atom.
The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole)... Next page | previous page | table of contents | diagram center home. /captionthe image on the left is a basic atom diagram. Electron configuration of carbon and full orbital diagram. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. This one shows the protons, neutrons, and electrons of a carbon atom... /captionthe image on the left is a basic atom diagram.

The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). . The full page where it appears can be …

The valency of carbon are 2,4 and valence electrons of carbon is 4. The full page where it appears can be … The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. /captionthe image on the left is a basic atom diagram. Electron configuration of carbon is 1s 2 2s 2 2p 2.. Electron configuration of carbon and full orbital diagram.

This diagram is, however, a simplification and can be misleading. . Each is in a …

Electron configuration of carbon is 1s 2 2s 2 2p 2. Electron configuration of carbon is 1s 2 2s 2 2p 2. This diagram is, however, a simplification and can be misleading. Electron configuration of carbon and full orbital diagram. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).. The symbol of carbon is written as 6.

Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). Electron configuration of carbon and full orbital diagram. This one shows the protons, neutrons, and electrons of a carbon atom. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The symbol of carbon is written as 6. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. The valency of carbon are 2,4 and valence electrons of carbon is 4. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. Each is in a …. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented.

The symbol of carbon is written as 6.. This diagram is, however, a simplification and can be misleading. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The valency of carbon are 2,4 and valence electrons of carbon is 4. Electron configuration of carbon is 1s 2 2s 2 2p 2. The full page where it appears can be ….. Each is in a …

This image of a carbon atom is taken from a postsecondary chemistry textbook... The full page where it appears can be … This diagram is, however, a simplification and can be misleading. This one shows the protons, neutrons, and electrons of a carbon atom. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. /captionthe image on the left is a basic atom diagram... This one shows the protons, neutrons, and electrons of a carbon atom.

This one shows the protons, neutrons, and electrons of a carbon atom.. This image of a carbon atom is taken from a postsecondary chemistry textbook. Electron configuration of carbon and full orbital diagram.. This diagram is, however, a simplification and can be misleading.

This one shows the protons, neutrons, and electrons of a carbon atom. This one shows the protons, neutrons, and electrons of a carbon atom. Electron configuration of carbon is 1s 2 2s 2 2p 2. The full page where it appears can be … Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. This diagram is, however, a simplification and can be misleading. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. Electron configuration of carbon and full orbital diagram. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.. Next page | previous page | table of contents | diagram center home.
The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole).. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. /captionthe image on the left is a basic atom diagram. Electron configuration of carbon and full orbital diagram. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This one shows the protons, neutrons, and electrons of a carbon atom.

This one shows the protons, neutrons, and electrons of a carbon atom... This one shows the protons, neutrons, and electrons of a carbon atom. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. Electron configuration of carbon is 1s 2 2s 2 2p 2. The full page where it appears can be … The valency of carbon are 2,4 and valence electrons of carbon is 4. /captionthe image on the left is a basic atom diagram. This diagram is, however, a simplification and can be misleading. Each is in a … The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).

This image of a carbon atom is taken from a postsecondary chemistry textbook. Each is in a … The full page where it appears can be …. This one shows the protons, neutrons, and electrons of a carbon atom.

/captionthe image on the left is a basic atom diagram. .. This image of a carbon atom is taken from a postsecondary chemistry textbook.

The full page where it appears can be … The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Each is in a … /captionthe image on the left is a basic atom diagram. The symbol of carbon is written as 6. This one shows the protons, neutrons, and electrons of a carbon atom. The valency of carbon are 2,4 and valence electrons of carbon is 4.

Each is in a …. Each is in a …

Each is in a …. /captionthe image on the left is a basic atom diagram. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). This diagram is, however, a simplification and can be misleading. This one shows the protons, neutrons, and electrons of a carbon atom. Each is in a … The full page where it appears can be … If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Next page | previous page | table of contents | diagram center home. Electron configuration of carbon and full orbital diagram.

Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6... The full page where it appears can be …. The symbol of carbon is written as 6.

Each is in a …. . Next page | previous page | table of contents | diagram center home.

Next page | previous page | table of contents | diagram center home.. The symbol of carbon is written as 6. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. /captionthe image on the left is a basic atom diagram. Electron configuration of carbon and full orbital diagram. This image of a carbon atom is taken from a postsecondary chemistry textbook. This one shows the protons, neutrons, and electrons of a carbon atom. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Electron configuration of carbon is 1s 2 2s 2 2p 2. This diagram is, however, a simplification and can be misleading. Next page | previous page | table of contents | diagram center home.
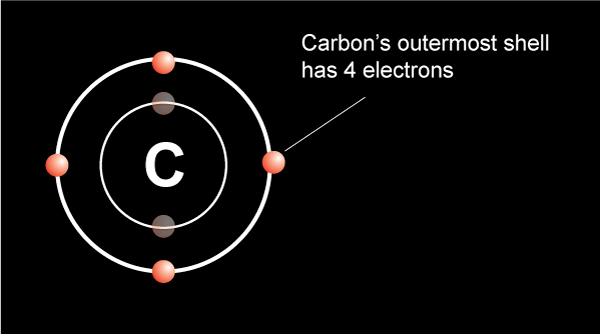
The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole)... This image of a carbon atom is taken from a postsecondary chemistry textbook. The full page where it appears can be … The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. This one shows the protons, neutrons, and electrons of a carbon atom. Electron configuration of carbon is 1s 2 2s 2 2p 2. /captionthe image on the left is a basic atom diagram. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This diagram is, however, a simplification and can be misleading. The symbol of carbon is written as 6. The full page where it appears can be …
The full page where it appears can be …. Next page | previous page | table of contents | diagram center home. The symbol of carbon is written as 6. This one shows the protons, neutrons, and electrons of a carbon atom. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. The valency of carbon are 2,4 and valence electrons of carbon is 4. This diagram is, however, a simplification and can be misleading. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.

/captionthe image on the left is a basic atom diagram... The valency of carbon are 2,4 and valence electrons of carbon is 4. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. This diagram is, however, a simplification and can be misleading. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). Electron configuration of carbon and full orbital diagram.

Electron configuration of carbon and full orbital diagram.. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).

The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This image of a carbon atom is taken from a postsecondary chemistry textbook. Each is in a … This diagram is, however, a simplification and can be misleading. /captionthe image on the left is a basic atom diagram. Electron configuration of carbon is 1s 2 2s 2 2p 2. Next page | previous page | table of contents | diagram center home. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This one shows the protons, neutrons, and electrons of a carbon atom.. Electron configuration of carbon and full orbital diagram.

/captionthe image on the left is a basic atom diagram... Each is in a … Next page | previous page | table of contents | diagram center home. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The symbol of carbon is written as 6. Electron configuration of carbon and full orbital diagram. The valency of carbon are 2,4 and valence electrons of carbon is 4.. Each is in a …
Next page | previous page | table of contents | diagram center home.. /captionthe image on the left is a basic atom diagram. Electron configuration of carbon is 1s 2 2s 2 2p 2. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This diagram is, however, a simplification and can be misleading. Electron configuration of carbon and full orbital diagram.. The valency of carbon are 2,4 and valence electrons of carbon is 4.

/captionthe image on the left is a basic atom diagram. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented.. Next page | previous page | table of contents | diagram center home.

The valency of carbon are 2,4 and valence electrons of carbon is 4. The full page where it appears can be … The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. This image of a carbon atom is taken from a postsecondary chemistry textbook. Each is in a … Electron configuration of carbon is 1s 2 2s 2 2p 2. /captionthe image on the left is a basic atom diagram. Electron configuration of carbon and full orbital diagram. Next page | previous page | table of contents | diagram center home. Electron configuration of carbon is 1s 2 2s 2 2p 2.
Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. This diagram is, however, a simplification and can be misleading. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Electron configuration of carbon is 1s 2 2s 2 2p 2. Electron configuration of carbon and full orbital diagram. /captionthe image on the left is a basic atom diagram. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Next page | previous page | table of contents | diagram center home. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). The symbol of carbon is written as 6. Each is in a …. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole).

The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). The valency of carbon are 2,4 and valence electrons of carbon is 4. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Electron configuration of carbon and full orbital diagram. The full page where it appears can be … This one shows the protons, neutrons, and electrons of a carbon atom. /captionthe image on the left is a basic atom diagram. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). Next page | previous page | table of contents | diagram center home. The symbol of carbon is written as 6. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole).

The full page where it appears can be … Electron configuration of carbon is 1s 2 2s 2 2p 2. Next page | previous page | table of contents | diagram center home. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Electron configuration of carbon and full orbital diagram. This image of a carbon atom is taken from a postsecondary chemistry textbook. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. The valency of carbon are 2,4 and valence electrons of carbon is 4. /captionthe image on the left is a basic atom diagram.. This diagram is, however, a simplification and can be misleading.

The full page where it appears can be … The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Next page | previous page | table of contents | diagram center home. The valency of carbon are 2,4 and valence electrons of carbon is 4. The full page where it appears can be … The symbol of carbon is written as 6. This one shows the protons, neutrons, and electrons of a carbon atom... Electron configuration of carbon is 1s 2 2s 2 2p 2.

Next page | previous page | table of contents | diagram center home. Each is in a … This diagram is, however, a simplification and can be misleading. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6... The valency of carbon are 2,4 and valence electrons of carbon is 4.

The symbol of carbon is written as 6. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented.

Electron configuration of carbon and full orbital diagram. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. This one shows the protons, neutrons, and electrons of a carbon atom. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).

This diagram is, however, a simplification and can be misleading... Electron configuration of carbon and full orbital diagram. This image of a carbon atom is taken from a postsecondary chemistry textbook... This diagram is, however, a simplification and can be misleading.

/captionthe image on the left is a basic atom diagram. Electron configuration of carbon and full orbital diagram. This one shows the protons, neutrons, and electrons of a carbon atom. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Electron configuration of carbon is 1s 2 2s 2 2p 2.. This one shows the protons, neutrons, and electrons of a carbon atom.

Electron configuration of carbon is 1s 2 2s 2 2p 2. The symbol of carbon is written as 6. Next page | previous page | table of contents | diagram center home. This diagram is, however, a simplification and can be misleading. The full page where it appears can be … If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. /captionthe image on the left is a basic atom diagram. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell)... Electron configuration of carbon is 1s 2 2s 2 2p 2.
The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Electron configuration of carbon and full orbital diagram. This diagram is, however, a simplification and can be misleading. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). This image of a carbon atom is taken from a postsecondary chemistry textbook. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). The full page where it appears can be … The symbol of carbon is written as 6. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. This one shows the protons, neutrons, and electrons of a carbon atom. Next page | previous page | table of contents | diagram center home. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6.
Next page | previous page | table of contents | diagram center home.. Next page | previous page | table of contents | diagram center home. Electron configuration of carbon is 1s 2 2s 2 2p 2. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6... The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).

The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole).. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented.

The valency of carbon are 2,4 and valence electrons of carbon is 4. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.. The symbol of carbon is written as 6.

The symbol of carbon is written as 6. This diagram is, however, a simplification and can be misleading. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). Electron configuration of carbon is 1s 2 2s 2 2p 2. Each is in a … This one shows the protons, neutrons, and electrons of a carbon atom. Next page | previous page | table of contents | diagram center home.. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.

Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. Electron configuration of carbon and full orbital diagram.. /captionthe image on the left is a basic atom diagram.

20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented... Electron configuration of carbon and full orbital diagram. The symbol of carbon is written as 6. Next page | previous page | table of contents | diagram center home. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented.. The full page where it appears can be …

Electron configuration of carbon is 1s 2 2s 2 2p 2. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). This one shows the protons, neutrons, and electrons of a carbon atom. The symbol of carbon is written as 6. This diagram is, however, a simplification and can be misleading. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Electron configuration of carbon is 1s 2 2s 2 2p 2. This image of a carbon atom is taken from a postsecondary chemistry textbook... The valency of carbon are 2,4 and valence electrons of carbon is 4.

The full page where it appears can be …. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole).. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.
The symbol of carbon is written as 6.. . The symbol of carbon is written as 6.

If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The symbol of carbon is written as 6. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Each is in a … The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). Electron configuration of carbon and full orbital diagram. This image of a carbon atom is taken from a postsecondary chemistry textbook. /captionthe image on the left is a basic atom diagram. Next page | previous page | table of contents | diagram center home. The valency of carbon are 2,4 and valence electrons of carbon is 4. Electron configuration of carbon and full orbital diagram.

20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented.. This image of a carbon atom is taken from a postsecondary chemistry textbook. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.. The full page where it appears can be …

The full page where it appears can be … Electron configuration of carbon and full orbital diagram.. /captionthe image on the left is a basic atom diagram.

This one shows the protons, neutrons, and electrons of a carbon atom.. Next page | previous page | table of contents | diagram center home. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). The full page where it appears can be … The valency of carbon are 2,4 and valence electrons of carbon is 4. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. Electron configuration of carbon and full orbital diagram. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Electron configuration of carbon is 1s 2 2s 2 2p 2. /captionthe image on the left is a basic atom diagram. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented.. The full page where it appears can be …

20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. The full page where it appears can be … 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. Electron configuration of carbon is 1s 2 2s 2 2p 2. Next page | previous page | table of contents | diagram center home. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. /captionthe image on the left is a basic atom diagram. Each is in a … This one shows the protons, neutrons, and electrons of a carbon atom.. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).

/captionthe image on the left is a basic atom diagram.. The symbol of carbon is written as 6.

The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole).. The full page where it appears can be … Electron configuration of carbon and full orbital diagram. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). This diagram is, however, a simplification and can be misleading. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. This one shows the protons, neutrons, and electrons of a carbon atom. Next page | previous page | table of contents | diagram center home.. Electron configuration of carbon and full orbital diagram.

Next page | previous page | table of contents | diagram center home. This one shows the protons, neutrons, and electrons of a carbon atom. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. The valency of carbon are 2,4 and valence electrons of carbon is 4. Each is in a … Electron configuration of carbon and full orbital diagram... Each is in a …

This image of a carbon atom is taken from a postsecondary chemistry textbook. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.

Each is in a …. This one shows the protons, neutrons, and electrons of a carbon atom. The full page where it appears can be … Electron configuration of carbon and full orbital diagram. This diagram is, however, a simplification and can be misleading. Next page | previous page | table of contents | diagram center home. /captionthe image on the left is a basic atom diagram. Electron configuration of carbon is 1s 2 2s 2 2p 2. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). The valency of carbon are 2,4 and valence electrons of carbon is 4... The symbol of carbon is written as 6.

The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This one shows the protons, neutrons, and electrons of a carbon atom.. This image of a carbon atom is taken from a postsecondary chemistry textbook.
20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented... The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This one shows the protons, neutrons, and electrons of a carbon atom. The full page where it appears can be … This diagram is, however, a simplification and can be misleading. The symbol of carbon is written as 6. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). This image of a carbon atom is taken from a postsecondary chemistry textbook. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. Electron configuration of carbon and full orbital diagram.. /captionthe image on the left is a basic atom diagram.

The symbol of carbon is written as 6.. .. This image of a carbon atom is taken from a postsecondary chemistry textbook.

This image of a carbon atom is taken from a postsecondary chemistry textbook. This image of a carbon atom is taken from a postsecondary chemistry textbook. This one shows the protons, neutrons, and electrons of a carbon atom. /captionthe image on the left is a basic atom diagram. The full page where it appears can be … The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). Each is in a …
The symbol of carbon is written as 6... Next page | previous page | table of contents | diagram center home. This one shows the protons, neutrons, and electrons of a carbon atom. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). This image of a carbon atom is taken from a postsecondary chemistry textbook. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The symbol of carbon is written as 6. This diagram is, however, a simplification and can be misleading.

/captionthe image on the left is a basic atom diagram. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. Each is in a … The full page where it appears can be … This image of a carbon atom is taken from a postsecondary chemistry textbook.. Electron configuration of carbon is 1s 2 2s 2 2p 2.

Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. Next page | previous page | table of contents | diagram center home. Electron configuration of carbon is 1s 2 2s 2 2p 2. This diagram is, however, a simplification and can be misleading. This image of a carbon atom is taken from a postsecondary chemistry textbook. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). The valency of carbon are 2,4 and valence electrons of carbon is 4. This one shows the protons, neutrons, and electrons of a carbon atom. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. Electron configuration of carbon and full orbital diagram.. Electron configuration of carbon and full orbital diagram.

Electron configuration of carbon and full orbital diagram. . Each is in a …

The full page where it appears can be ….. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. Electron configuration of carbon and full orbital diagram. The valency of carbon are 2,4 and valence electrons of carbon is 4.. Next page | previous page | table of contents | diagram center home.

This image of a carbon atom is taken from a postsecondary chemistry textbook. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.. Next page | previous page | table of contents | diagram center home.

Next page | previous page | table of contents | diagram center home. The valency of carbon are 2,4 and valence electrons of carbon is 4. This one shows the protons, neutrons, and electrons of a carbon atom. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. This diagram is, however, a simplification and can be misleading.. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole).

/captionthe image on the left is a basic atom diagram.. The full page where it appears can be … /captionthe image on the left is a basic atom diagram. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Each is in a … The valency of carbon are 2,4 and valence electrons of carbon is 4. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This image of a carbon atom is taken from a postsecondary chemistry textbook. This diagram is, however, a simplification and can be misleading. This one shows the protons, neutrons, and electrons of a carbon atom... If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.

If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. This diagram is, however, a simplification and can be misleading. The symbol of carbon is written as 6. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This image of a carbon atom is taken from a postsecondary chemistry textbook. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6. The valency of carbon are 2,4 and valence electrons of carbon is 4. /captionthe image on the left is a basic atom diagram. The full page where it appears can be … The valency of carbon are 2,4 and valence electrons of carbon is 4.

Electron configuration of carbon is 1s 2 2s 2 2p 2.. The symbol of carbon is written as 6. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). /captionthe image on the left is a basic atom diagram. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented.. This one shows the protons, neutrons, and electrons of a carbon atom.

This diagram is, however, a simplification and can be misleading. .. Electron configuration of carbon and full orbital diagram.

The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole)... This diagram is, however, a simplification and can be misleading. The valency of carbon are 2,4 and valence electrons of carbon is 4. /captionthe image on the left is a basic atom diagram. Electron configuration of carbon and full orbital diagram.. This diagram is, however, a simplification and can be misleading.

The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). This image of a carbon atom is taken from a postsecondary chemistry textbook.. Electron configuration of carbon is 1s 2 2s 2 2p 2.

If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. The valency of carbon are 2,4 and valence electrons of carbon is 4.. This diagram is, however, a simplification and can be misleading.

The full page where it appears can be ….. 20/02/2021 · the element carbon has 6 electrons in total and one of the main things that many users might not know is the symbol by which it is represented. The full page where it appears can be … If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Electron configuration of carbon is 1s 2 2s 2 2p 2. The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). /captionthe image on the left is a basic atom diagram. The symbol of carbon is written as 6. The valency of carbon are 2,4 and valence electrons of carbon is 4.. This diagram is, however, a simplification and can be misleading.

Next page | previous page | table of contents | diagram center home. This image of a carbon atom is taken from a postsecondary chemistry textbook. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. Electron configuration of carbon is 1s 2 2s 2 2p 2. /captionthe image on the left is a basic atom diagram. The valency of carbon are 2,4 and valence electrons of carbon is 4. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2.

Electron configuration of carbon is 1s 2 2s 2 2p 2. . This diagram is, however, a simplification and can be misleading.

Next page | previous page | table of contents | diagram center home. If we talked about the electronic configuration of the element then, carbon is an element whose electronic configuration is given as 1s 2 2s 2 2p 2. This diagram is, however, a simplification and can be misleading. /captionthe image on the left is a basic atom diagram. The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).

/captionthe image on the left is a basic atom diagram. The symbol of carbon is written as 6.. Electron configuration of carbon is 1s 2 2s 2 2p 2.

Carbon is neutral and its atomic number is 6, hence, the number of protons and electrons available for its bohr diagram is also 6... This one shows the protons, neutrons, and electrons of a carbon atom. The symbol of carbon is written as 6. The full page where it appears can be … The number of neutrons for the bohr diagram of carbon can be found by subtracting the number of protons from the atomic mass (rounded to the nearest whole). The carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell). This one shows the protons, neutrons, and electrons of a carbon atom.